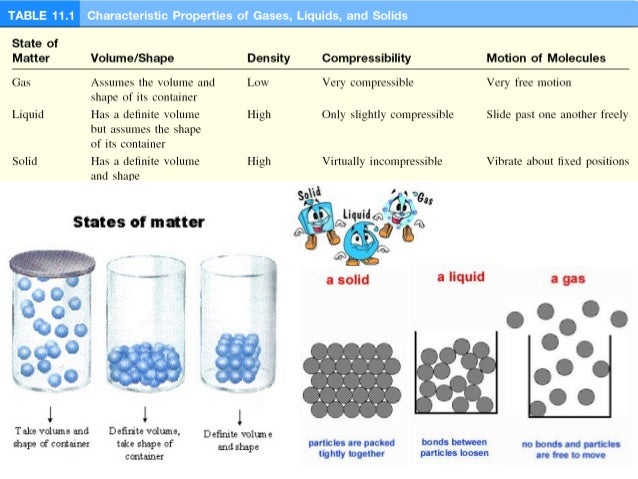
What is meant by van der Waals contact distance? The Van der Waals interaction is the weakest of all the intermolecular forces that hold molecules together. What causes van der Waals forces? Induced electrical interactions between two or more atoms or molecules that are very near to each other produce Van der Waals forces. Van Der Waals Bonding is the form of bonding seen in N2 molecules. Van der Waals interaction is the weakest of all intermolecular attractions between molecules. Van der Waals forces are driven by induced electrical interactions between two or more atoms or molecules that are very close to each other. Here is a chart to compare the relative weakness of Van der Waals forces to other intermolecular attractions. However, with a lot of Van der Waals forces interacting between two objects, the interaction can be very strong. Is van der Waals interaction strong or weak? To learn more about Van der Waals forces and other related concepts (such as dipole moment ), register with BYJU S and download the mobile application on your smartphone. London forces are attractive forces arising due to instantaneous dipoles. 
These forces depend on the polarizability of a molecule or an atom. The molecular forces between non-polar molecules are called London dispersion forces. These Van Der Waals interactions are stronger than Dispersion forces but weaker than ion-ion interactions. What is the difference between dispersion forces and Van der Waals forces? To learn more about Van der Waals forces and other related concepts (such as dipole moment ), register with BYJUS and download the mobile application on your smartphone. The interaction increases when molecules are closer. Van Der Waals forces are not directional. They are weaker than ionic, covalent bonds. What is the difference between ionic and Van der Waals forces? It is different from covalent or ionic bonds and is not a resultant attraction of chemical, electronic bonds. 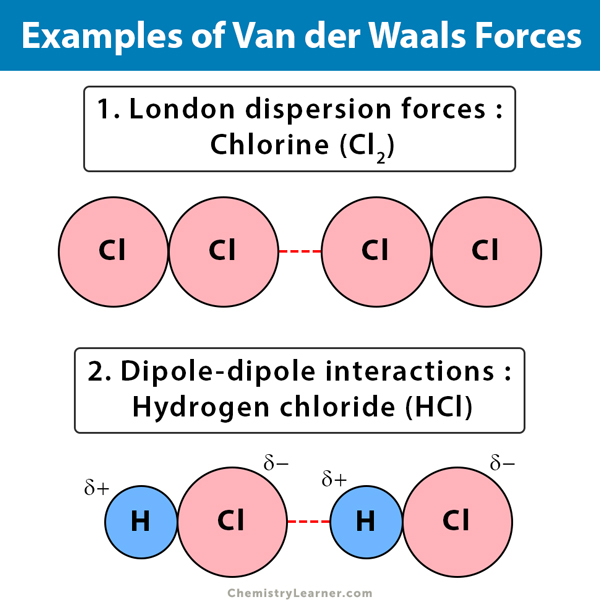
In chemistry, van der Waal force is a kind of interaction which is dependent on the distance between molecules or atoms. These forces are independent of temperature. 
Following are the characteristics of Van Der Waal forces. What are the characteristics of Van der Waal forces?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
